In chemistry, irreversible reactions receive all the attention: the well-represented fires and explosions on YouTube. However, not all reactions reach completion. Sometimes, the products of a chemical reaction can react with one another to regenerate the starting materials, resulting in a reversible chemical reaction. These are the types of reactions that occur all around us and are so commonplace that we never notice them. A mind-boggling number of protons in my Diet Coke (pH 3.39) are exchanging places with water molecules at any given time, but since there is no net change, the process is virtually invisible.
As the final topic in "From General Chemistry to Organic Chemistry," we will discuss chemical equilibrium, a major topic in organic chemistry. This concept extends beyond chemical reactions to a variety of processes, such as how substrates bind to enzymes and how molecules can change their three-dimensional structure reversibly.
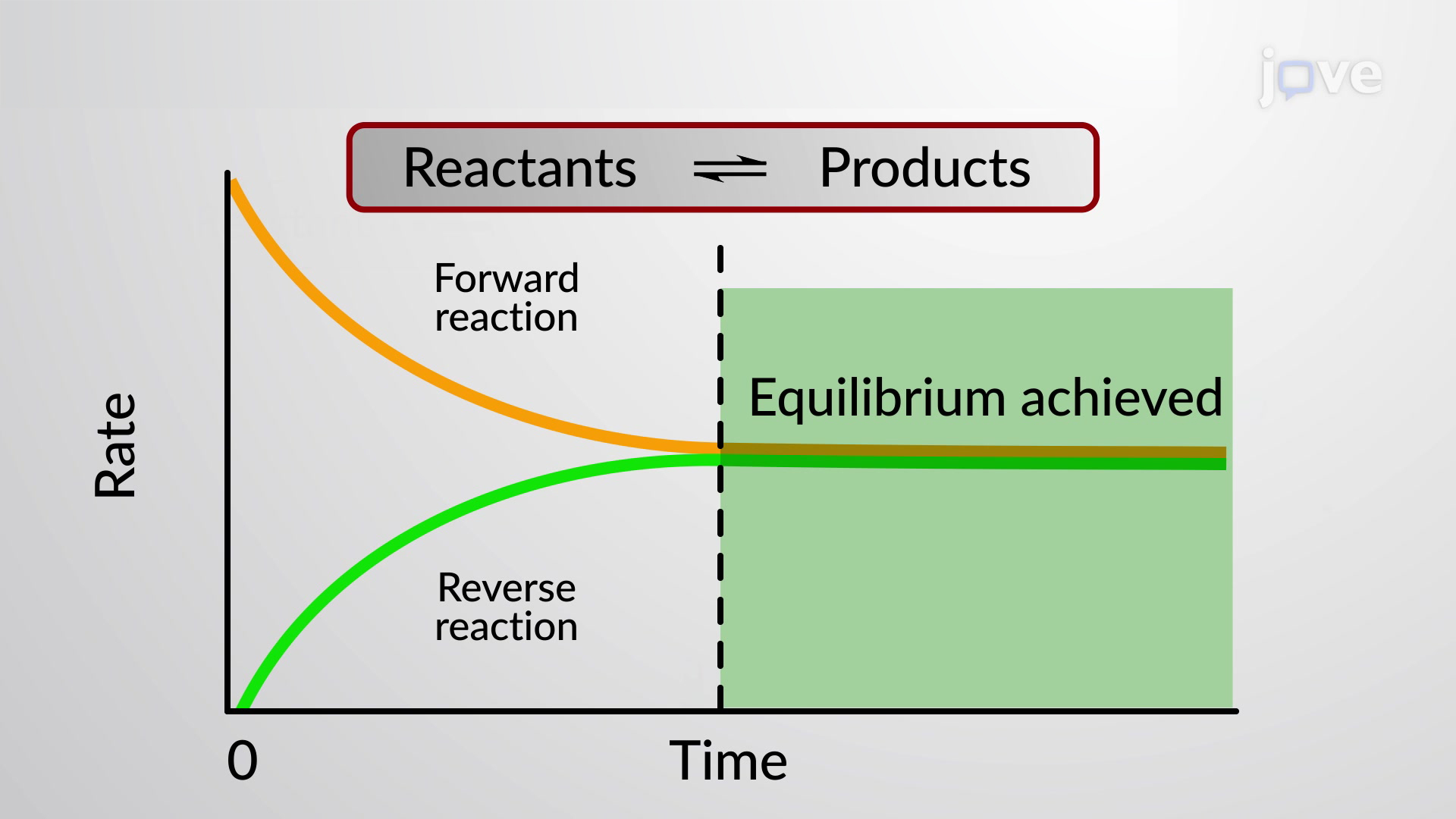
So, what exactly is chemical equilibrium? Why is it essential? What does it mean to say that chemical equilibrium has been reached?
Let's begin with the fundamentals:
A chemical equilibrium can only exist when a significant reverse reaction is present.
When chemical equilibrium is reached, the concentrations of reactants and products do not change over time.
At equilibrium, the process's delta G is equal to zero. No driving force exists.
In General Chemistry, acid-base reactions likely serve as the most significant introduction to equilibrium. The reaction between the strong acid hydrochloric acid (HCl) and water, which produces H3O(+), is essentially irreversible. The weak acid acetic acid (CH3COOH) reacts with H2O to produce H3O(+) and CH3COO(–), although the opposite reaction is also possible.
It is important to note that the value of this equilibrium constant measures the relative strengths of acids. This yields the extremely useful pKa value, which is equal to the negative logarithm of the Ka value for this reaction. pKa is an excellent method for comparing the relative acidity of various molecules. As molecule acidity increases, this equilibrium will shift further and further to the right. Strong acids have low pKas (just as strong acids dissolved in water result in low pH) and weak acids have high pKas.
You probably don't consider it, but chemical reactions in equilibrium are constantly occurring in such subtle ways that you don't even notice them. In each of the cans in the nearby soda machine, for example, equilibria have been established between water and the (pressurised) carbon dioxide dissolved in solution, which forms carbonic acid; at the same time, carbonic acid molecules are dissociating to release carbon dioxide and water.
Change in carbon dioxide, water, and carbonic acid concentrations: zero.
G can be determined from the equilibrium constant.
At equilibrium, no driving force exists. There is no G for the reaction. We can solve for G (if we know the equilibrium constant) or the equilibrium constant (if we know G) using the Gibbs free energy isotherm equation, which is a very useful numerical relationship between the equilibrium constant and G.
The Gibbs equation: G = –RT log K
In your organic chemistry course, you will encounter numerous examples of reversible reactions that result in mixtures. [One specific example is in the study of conformations, which are the three dimensional states of molecules formed through the reversible rotation of bonds]. This is how to use the equation to determine G, given K:
You can even plug this equation into a graph to visualise the relationship (if you'll excuse my terrible Excel skills).
Consider how steep the initial portion of this curve is! It's remarkable how small the energy difference is for a reaction that produces a 9:1 ratio of products. Compare it to the strength of the C-H bond, which is 430 kJ/mol, and you'll realise that 5.4 kJ/mol is not very much. For a reaction to produce a 99:1 ratio, the energy difference must be just 11.4 kJ/mol.
Here is the final important concept regarding equilibrium. The science of organic chemistry is an applied one. Frequently, we are concerned with transforming one substance into another. Frequently, we want to halt the reverse reaction to increase our product yields.
Frequently, we are able to manipulate the equilibrium to obtain the products we desire. This is commonly described as driving the reaction forward. This is accomplished by removing one of the byproducts of the forward reaction, thereby preventing the occurrence of the reverse reaction.
Here is an illustration. When an aldehyde is combined with an amine, an imine and a molecule of water are produced. This reaction is reversible, as water can react with imine to produce the original product.
Here is the central point. If you can find a way to remove the water, you will effectively stop k2 (the opposite reaction). What effect will that have on the overall reaction? It will continue to move forward (to the right) until equilibrium is restored. Which is never, as the desiccant has been added. One product instead of a mixture is the result.
Several practical methods exist for removing water, including the addition of a substance (called a dessicant) that reacts irreversibly with water and thus removes it from the equation. One example is magnesium sulphate (MgSO4), which forms a hydrate.
This principle should be familiar to you; it is known as Le Chatelier's principle.
In organic chemistry, you will encounter many examples of Le Chatelier's principle, as there are many reversible reactions (some in Org 1, but many in Org 2).
If you understand a reaction and its products, you can select conditions that permit the removal of products, thereby preventing an equilibrium from forming and driving the reaction towards the desired products.
Post a Comment